Facts about Density

The average density of an object equals its total mass divided by its total volume.

Water's density can also be measured as nearly one gram per cubic centimeter (at maximum density) in non-SI units.

The superscripts indicate the temperature at which the density of the material is measured, and the subscripts indicate the temperature of the water to which it is compared.

Relative density is often used by geologists and mineralogists to help determine the mineral content of a rock or other sample.

The singularity at the centre of a black hole, according to general relativity, does not have any volume, so its density is undefined.

Density in terms of the SI base units is expressed in kilograms per cubic meter (kg/m3).

Relative density is more easily and perhaps more accurately measured without measuring volume.

Specific gravity of Biogas== The density of biogas at 50% methane proportion is 1.227 kg/m3.

The relative density result is the dry sample weight divided by that of the displaced water.

Sometimes when this is done, it is a comparison of the density of the commodity being measured at that temperature, with the density of water at the same temperature.
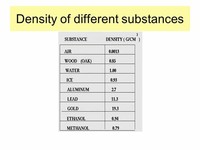
Density will determine the "order" in which each substance will appear in a bottle.

The maximum density of pure water at a pressure of one standard atmosphere is 999.861kg/m3; this occurs at a temperature of about 3.98 °C (277.13 K).

Naturally, a lesser volume of liquid provided for urination with the same amount of toxins would result in a higher specific gravity—a higher density of the solutes.

The reason that relative density is measured in terms of the density of water is because that is the easiest way to measure it in the field.

Experimentally density can be found by measuring the dry weight ( ), the wet weight ( ) and submersed weight ( ), usually in water.

Note too that fashion can change to enforce uniformity, as in the case where so-called "Mao suits" became the national uniform of China.

The relative density therefore has nearly the same value as density of the material expressed in grams per cubic centimeter, but without any units of measurement.

Perhaps the highest density known is reached in neutron star matter (neutronium).

From 1901 to 1964, a litre was defined as exactly the volume of 1 kg of water at maximum density, and the maximum density of pure water was 1.000 000 kg/L (now 0.999 972 kg/L).

Under specified conditions of temperature and pressure, the density of a fluid is defined as described below.

In both types of bulk density, some of the volume is taken up by the spaces between the grains of sand.



