Facts about Gasoline

Like other alkenes, gasoline burns in the vapor phase and, coupled with its volatility, this makes leaks highly dangerous when sources of ignition are present.

Gasoline is rather volatile (meaning it readily evaporates), requiring that storage tanks on land and in vehicles be properly sealed.

In Brazil, the Brazilian National Agency of Petroleum, Natural Gas and Biofuels (ANP) requires that gasoline for automobile use has 23 percent of ethanol added to its composition.

A common ethanol-gasoline mix of 10 percent ethanol mixed with gasoline is called gasohol or E10, and an ethanol-gasoline mix of 85% ethanol mixed with gasoline is called E85.

Initial marketing efforts will focus on an additive package for converting ordinary Biodiesel into gasoline, adding the Biolene additive package.

The various refinery streams blended together to make gasoline all have different characteristics.

Possession and use of leaded gasoline in a regular on-road vehicle now carries a maximum $10,000 fine in the US.

The bulk of a typical gasoline consists of hydrocarbons with between 5 and 12 carbon atoms per molecule.

The U.S. accounts for about 44 percent of the world’s gasoline consumption.

Based on externalities, some countries, for example, in Europe and Japan, impose heavy fuel taxes on fuels such as gasoline.

Overall a typical gasoline is predominantly a mixture of paraffins (alkanes), naphthenes (cycloalkanes), and olefins (alkenes).

Gasoline originally referred to any liquid used as the fuel for a gasoline-powered engine, other than diesel fuel or liquefied gas; methanol racing fuel would have been classed as a type of gasoline.

The mixture known as gasoline, when used in high compression internal combustion engines, has a tendency to autoignite(detonation) causing a damaging "engine knocking" (also called "pinging") noise.

Gasoline was also used in kitchen ranges and for lighting, and is still available in a highly purified form, known as camping fuel or white gas, for use in lanterns and portable stoves.
In 2003, The U.S. consumed Template:Convert/GL, which equates to 1.3 gigalitres of gasoline each day (about 360 million U.S. gallons or 300 million imperial gallons).

Many of these alternatives are less damaging to the environment than gasoline, but the first generation biofuels are still not 100 percent clean.

The ban on leaded gasoline led to thousands of tons of lead not being released in the air by automobiles.

Currently many countries set tight limits on gasoline aromatics in general, benzene in particular, and olefin (alkene) content.

In 1995, leaded fuel accounted for only 0.6 percent of total gasoline sales and less than 2,000 short tons of lead per year.

The Material Safety Data Sheet for unleaded gasoline shows at least fifteen hazardous chemicals occurring in various amounts.

Chevron, 76, Shell, and several other brands market ethanol-gasoline blends.

Through misuse as an inhalant, gasoline also contributes to damage to health.
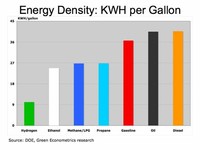
Energy content per kilogram is higher than for gasoline (higher hydrogen to carbon ratio).

Different countries have some variation in what RON (Research Octane Number) is standard for gasoline, or petrol.

Biogasoline has the advantage of not needing any change in vehicle or distribution infrastructure.

The resulting fuel is often known as reformulated gasoline (RFG) or oxygenated gasoline.

Gasoline, as delivered at the pump, also contains additives to reduce internal engine carbon buildups, improve combustion, and to allow easier starting in cold climates.

Gasoline contains about 34.8 MJ/L or 132 MJ/US gallon.

Gasoline containing acidic contaminants such as naphthenic acids can be addressed with additives including strongly basic organo-amines such as N,N-diethylhydroxylamine, preventing metal corrosion and breakdown of other antioxidant additives due to acidity.

Leaded gasoline was phased out in sub-Saharan Africa effective January 1, 2006.

The desired volatility depends on the ambient temperature: in hotter climates, gasoline components of higher molecular weight and thus lower volatility are used.

The weight-density of gasoline is about 740 kg/mі (6.175 lb/US gal; 7.416 lb/imp gal).

Mogas, short for motor gasoline, distinguishes automobile fuel from aviation gasoline, or avgas.

The U.S. used about 510 billion liters (138 billion U.S. gal/115 billion imp gal) of gasoline in 2006, of which 5.6 percent was mid-grade and 9.5 percent was premium grade.

The usage derives from the chemical benzene, not from Bertha Benz, who used chemists' shops to purchase the gasoline for her famous drive from Mannheim to Pforzheim in 1888.
The name gasoline is similar to that of other petroleum products of the day, most notably petroleum jelly, a highly purified heavy distillate, which was branded Vaseline.

Naphthenes, olefins, and aromatics increase the octane rating of the gasoline whereas the n-paraffins have the opposite effect.

The chief risks of such leaks come not from vehicles, but from gasoline delivery truck accidents and leaks from storage tanks.

By the early 1930s, automobile gasoline had an octane reading of 40 and aviation gasoline of 75-80.

Gummy, sticky resin deposits result from oxidative degradation of gasoline.

Gasoline is more volatile than diesel oil, Jet-A or kerosene, not only because of the base constituents, but because of the additives that are put into it.

Gasoline can also contain some other organic compounds: Such as organic ethers (deliberately added), plus small levels of contaminants, in particular sulfur compounds such as disulfides and thiophenes.

Volatility standards may be relaxed (allowing more gasoline components into the atmosphere) during emergency anticipated gasoline shortages.

The shortened form "gas" for gasoline was first recorded in American English in 1905 and is often confused with the older words gas and gases that have been used since the early 1600s.

A growing number of countries have drawn up plans to ban leaded gasoline in the near future.

When gasoline is left for a period of time, gums and varnishes may build up and precipitate in the gasoline, causing "stale fuel."

Many of the non-aliphatic hydrocarbons naturally present in gasoline (especially aromatic ones like benzene), as well as many anti-knocking additives, are carcinogenic.

On average, about 19.5 US gallons (16.2 imp gal/74 L) of gasoline are available from a Template:Convert/LoffAoffDbSonUSre barrel of crude oil (about 46 percent by volume), varying due to quality of crude and grade of gasoline.

Before dedicated filling stations were established, early motorists bought gasoline in cans to fill their tanks.

Before gasoline was used as fuel for engines, it was sold in small bottles as a treatment against lice and their eggs.

The main reason for the lower energy content (per litre) of LPG in comparison to gasoline is that it has a lower density.

Aviation gasoline with such high octane numbers could only be refined through a process of distillation of high-grade petroleum.

Companies such as Sapphire Energy are developing a means to "grow" gasoline, that is, produce it directly from living organisms (that is, algae).

In British English "gasoline" can refer to a different petroleum derivative historically used in lamps, but this is now uncommon.







