Facts about Iron
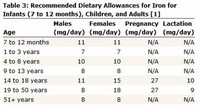
The Recommended Dietary Allowance (RDA) for iron varies considerably based on the age, gender, and source of dietary iron (heme-based iron has higher bioavailability).

Humans experience iron toxicity above 20 milligrams (mg) of iron for every kilogram (kg) of weight, and 60 mg/kg is a lethal dose.

One element of the medical approach is a specific agent called deferoxamine, used to bind and expel excess iron from the body in case of iron toxicity.

We are now familiar with many materials that are stronger and sturdier than iron, such as diamond or steel.

When the body is fighting a bacterial infection, the body sequesters iron inside of cells (mostly in the storage molecule ferritin), so that it cannot be used by bacteria.

Blood donors are at special risk of low iron levels and are often advised to supplement their iron intake.

Cast iron development lagged in Europe, as the smelters could achieve temperatures of only about 1000 °C.

Inorganic iron involved in redox reactions is also found in the iron-sulfur clusters of many enzymes, such as nitrogenase (involved in the synthesis of ammonia from nitrogen and hydrogen) and hydrogenase.

Iron and its alloys, however, tend to rust and need to be protected by such methods as painting, galvanization, or the application of plastic coatings.

Iron becomes toxic when it exceeds the amount of transferrin present to bind free iron.

Iron is used in the production of steel, an alloy of different metals and some nonmetals, particularly carbon.

In any event, by the late fourteenth century, a market for cast iron goods began to form, as a demand developed for cast iron cannonballs.

The main drawback in using iron and its alloys is that they suffer from rust if not protected in some way.

Early iron smelting used charcoal, as both the heat source and reducing agent.

Painting, galvanization, plastic coating, and bluing are some techniques used to protect iron from rust by excluding water and oxygen, or by sacrificial protection.

The most bioavailable form of iron supplement (ten to fifteen times more bioavailable than any other) is iron amino acid chelate.

The Earth's core is believed to consist largely of a metallic iron-nickel alloy.

To obtain elemental iron, the impurities must be removed by chemical reduction.

Of all metals, iron makes up the highest level of production and usage.

The vast majority of Chinese iron manufacture, from the Zhou dynasty onward, was of cast iron.

Iron, cobalt, and nickel have a number of similar properties and were once grouped together as group 8B.

Concurrent with the transition from bronze to iron was the discovery of carburization, which was the process of adding carbon to the irons of the time.

Iron ore is produced in 48 countries, of which the five largest producers are China, Brazil, Australia, Russia, and India, accounting for 70 percent of world iron ore production.

Iron and nickel are notable for being the final elements produced by nuclear fusion reactions within stars—processes that are collectively known as stellar nucleosynthesis.

to 1200 B.C.E., iron was used increasingly in the Middle East, but did not supplant the dominant use of bronze.

To say that iron is free does not mean that it is free floating in bodily fluids.

Iron is one of the most common elements on Earth, making up about 5 percent of the Earth's crust.

Through a good portion of the Middle Ages, in Western Europe, iron was thus still being made by the working of iron blooms into wrought iron.
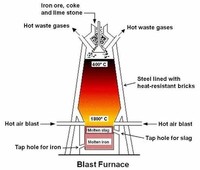
Producing blast furnaces capable of temperatures exceeding 1300 K, the Chinese developed the manufacture of cast iron or pig iron.

Scientists think that iron (and other elements) on Earth were once part of the material ejected by a supernova (exploding star).

The concentration of iron in the various layers of the Earth ranges from very high at the inner core to only a few percent in the outer crust.

Iron is also used for making magnets, and iron(III) oxide in particular is used for the manufacture of magnetic storage media for computers.

Iron overload disorders are based on a genetic inability to regulate iron uptake.

The famous iron pillar at the Qutb complex in Delhi is made of very pure (98 percent) iron and has not rusted or eroded till this day.

Iron-containing enzymes help transport various soluble gases (including oxygen and carbon dioxide) and catalyze oxidation reactions in living organisms.

Most of this iron is found in various iron oxides, such as the minerals hematite, magnetite, and taconite.

Many people have a genetic susceptibility to iron overload without realizing it and without knowing a family history of the problem.

Iron is a ductile, malleable, gray-colored metal and is a good conductor of heat and electricity.

Approximately 1,100 Mt (million tons) of iron ore was produced in the world in 2000, with a gross market value of approximately 25 billion US dollars.

Perhaps they did not want hotter temperatures, as they were seeking to produce blooms as a precursor of wrought iron, not cast iron.

Wrought iron was very low in carbon content and was not easily hardened by quenching.

Excessive iron is toxic to humans because excess ferrous iron reacts with peroxides in the body, producing free radicals.

Iron is believed to be the tenth most abundant chemical element in the universe.

Iron was recovered as sponge iron, a mix of iron and slag with some carbon and/or carbide.
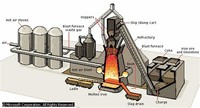
Iron is extracted from iron ore and is almost never found in the free elemental state.

Over-consumption of iron, often the result of children consuming large quantities of ferrous sulfate tablets intended for adults, is the most common toxicological cause of death in children under six.

Still, the "iron" metaphor persists among many cultures, apparently rooted in the Iron Age, when iron defeated bronze.

The first signs of use of iron come from artifacts of the Sumerians and Egyptians dated to around 4000 B.C.E.

Classical authors ascribe the first invention of ironsmithing to peoples of the Caucasus and eastern Anatolia, such as the Khaldi (Chaldei) and the Khalib (Chalybes).

to 2000 B.C.E., increasing numbers of smelted iron objects (distinguishable from meteoric iron by the lack of nickel in the product) appear in Mesopotamia, Anatolia, and Egypt.

The word "iron" is used metaphorically as an adjective, to describe certain iron-like traits, including sturdy, strong, stern, harsh, unyielding, inflexible, rigid, or robust.

Some of the earliest casting of iron in Europe occurred in Sweden, at two sites, Lapphyttan and Vinarhyttan, between 1150 and 1350 C.E.

Some in ancient Greece considered the Halybes to be "the inventors of iron."

Iron provided by dietary supplements is often found as iron(II) fumarate.

The use of iron metal filings in organic syntheses is mainly for the reduction of nitro compounds.

below) suggest that iron was then being generated as a byproduct of copper refining, as sponge iron, and was not reducible by the metallurgy of the time.

Too much ingested iron can damage the cells of the gastrointestinal tract directly and may enter the bloodstream by damaging the cells that would otherwise regulate its entry.

The critical factor in this transition does not appear to be the sudden onset of a superior iron working technology, but disruption of the supply of tin.

Iron uptake is tightly regulated by the human body, which has no physiologic means of excreting iron and regulates iron solely by regulating uptake.

Iron distribution is heavily regulated in mammals, both as a defense against bacterial infection and because of the potential biological toxicity of iron (noted below).

Industrially, it is produced starting from iron ores, principally hematite (nominally Fe2O3) and magnetite (Fe3O4) by reducing the ore with carbon in a blast furnace at temperatures of about 2,000 °C.

Naturally occurring iron consists of four isotopes: 5.85 percent of slightly radioactive 54Fe (half-life >3.1Ч1022 years), 91.75 percent of stable 56Fe, 2.12 percent of stable 57Fe, and 0.28 percent of stable 58Fe.

Some of the various alloys of iron and their applications are given below.

Much of the past work on measuring the isotopic composition of iron centered on determining 60Fe variations due to processes accompanying nucleosynthesis (that is, through meteorite studies) and ore formation.

Some have linked the iron in meteorites to a verse in the Quran (57:25) that says, "… and We sent down iron in which is incredible strength and many benefits for mankind."

In China, the first irons used were also meteoric iron, with archaeological evidence for items made of wrought iron appearing in the northwest, near Xinjiang, in the eighth century B.C.E.

Good sources of dietary iron include meat, fish, poultry, lentils, beans, leaf vegetables, tofu, chickpeas, black-eyed peas, strawberries, and farina.

Iron production constitutes about 95 percent of the tonnage of all metals produced worldwide.

The iron absorbed from the duodenum binds to transferrin and is carried by blood to different cells.

In any event, by the late fourteenth century, a market for cast iron goods began to form, as a demand developed for cast iron cannonballs.
Most of it is used to make steel — an alloy of iron and carbon — which is in turn used in manufacturing and civil engineering, for instance, to make reinforced concrete. Stainless steel, which contains at least 10.5 percent chromium, is highly resistant to corrosion.Aug 22, 2017
Earth's most important iron ore deposits are found in sedimentary rocks. They formed from chemical reactions that combined iron and oxygen in marine and fresh waters. The two most important minerals in these deposits are iron oxides: hematite (Fe2O3) and magnetite (Fe3O4).
Iron is the sixth most common element in the universe. Iron is the fourth most abundant element in the Earth's crust by weight. It is the most abundant element in the total composition of the planet. Iron is found in both the inner and outer core of Earth.
Iron makes up about 5% of the Earth's crust and a large part of the Earth's core. Iron also comes from meteorites. On earth Iron is found mainly inside the minerals hematite (Fe2O3) and magnetite (Fe3O4.) Removing the iron from these minerals is a reasonably long process involving many large industrial factories.
Iron is a mineral that is naturally present in many foods, added to some food products, and available as a dietary supplement. Iron is an essential component of hemoglobin, an erythrocyte protein that transfers oxygen from the lungs to the tissues [1]. ... Dietary iron has two main forms: heme and nonheme [1].Feb 11, 2016
Iron is an important component of hemoglobin, the substance in red blood cells that carries oxygen from your lungs to transport it throughout your body. ... If you don't have enough iron, your body can't make enough healthy oxygen-carrying red blood cells. A lack of red blood cells is called iron deficiency anemia.
Iron is an important cofactor required for a number of essential cell functions and hence is a vital nutrient. However, iron can also be dangerous as a catalyst of free radical reactions. Accordingly, intracellular iron homeostasis and body iron balance are tightly regulated.
Hemoglobin is a protein in red blood cells that transports oxygen to tissues in the body. Myoglobin, the compound that carries oxygen to the muscle cells, also requires iron. In addition, iron is involved in reactions within the body that produce energy.
Iron in diet. Iron is a mineral found in every cell of the body. Iron is considered an essential mineral because it is needed to make hemoglobin, a part of blood cells.Jan 7, 2017
Magnets attract iron due to the influence of their magnetic field upon the iron. ... When exposed to the magnetic field, the atoms begin to align their electrons with the flow of the magnetic field, which makes the iron magnetized as well. This, in turn, creates an attraction between the two magnetized objects.Mar 22, 2009
Since iron is a ferromagnetic material, a magnetic field induces each particle to become a tiny bar magnet. The south pole of each particle then attracts the north poles of its neighbors, and this process repeated over a wide area creates chains of filings parallel to the direction of the magnetic field.








