Facts about Ytterbium

Of interest to modern quantum optics, the different ytterbium isotopes follow either Bose-Einstein statistics or Fermi-Dirac statistics, leading to interesting behavior in optical lattices.

The chemical and physical properties of ytterbium could not be determined until 1953 when the first nearly pure ytterbium was produced.

Ytterbium compounds are, however, known to cause skin and eye irritation and may be teratogenic.

All compounds of ytterbium should be treated as highly toxic although initial studies appear to indicate that the danger is limited.

Ytterbium was discovered by the Swiss chemist Jean Charles Galissard de Marignac in 1878.

Ytterbium is found with other rare earth elements in several rare minerals.

The isotopes of ytterbium range in atomic weight from 150.955 u (Yb-151) to 179.952 u (Yb-180).

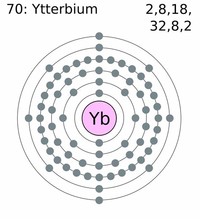
Ytterbium (chemical symbol Yb, atomic number 70) is a soft silvery metallic rare earth element.
Naturally occurring ytterbium is composed of seven stable isotopes—Yb-168, Yb-170, Yb-171, Yb-172, Yb-173, Yb-174, and Yb-176—with Yb-174 being the most abundant (31.8 percent natural abundance).

Neoytterbia would later become known as the element ytterbium and lutecia would later be known as the element lutetium.

Known compounds of ytterbium are rare—they haven't been well characterized yet.

Ytterbium is an inner transition metal (or lanthanide) that lies in period six of the periodic table, between thulium and lutetium.

Ytterbium is normally difficult to separate from other rare earths but ion-exchange and solvent extraction techniques developed in the late twentieth century have simplified separation.


