Does hot water have more density than cold water?
Best Answers
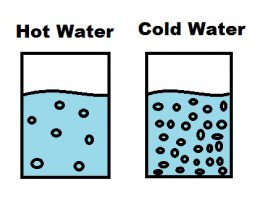
Cold water has greater density than hot because its water molecules are more sluggish; vibrations and movements are slower and less energetic. The molecules bounce and jostle each other less, so more can fit together in a smaller space. read more
Source: sciencing.com
Cold water is always more dense than warm water; the density change amounts to about 4 tenths of one percent between near-freezing and 30 degrees Celsius (86 degrees Fahrenheit). Although small, the difference allows warm water to "float" on top of cold water, a phenomenon that occurs every day in the world's oceans. read more
Source: sciencing.com
Encyclopedia Research
Wikipedia:
Related Questions
Related Facts
Related Types
Image Answers
Source: sadie14.hubpages.com
Further Research
Hot water is heavier than cold water.
www.physicsforums.com
Properties of the sea: salinity and temperature
www.learnz.org.nz
Temperature and Density
www.middleschoolchemistry.com
Why Is Hot Water Less Dense Than Cold Water?
sciencing.com

