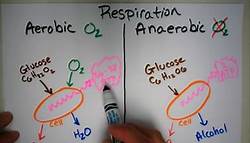
How efficient is cellular respiration?
Best Answers
The simplified reaction is C6H12O6 + 6 O2 = 6 CO2 + 6 H2O. The reaction is also exothermic, meaning it can happen spontaneously and releases energy in the form of heat. So thermodynamically this is very efficient. read more
One can easily determine the energy efficiency of cellular respiration by calculating the standard free energy change, a thermodynamic quantity, between the reactants and products. On this basis, biochemists often quote the overall efficiency of cellular respiration as about 40%, with the additional 60% of the energy given off as heat. read more
Source: science.jrank.org
The end product you are looking for determines the numerator of your efficiency calculation. read more
Encyclopedia Research
Wikipedia:
Related Questions
Related Facts
Related Types
Image Answers

Source: slideplayer.com
Further Research
Display a local file in Google Maps
science.jrank.org
EFFICIENCY OF ATP PRODUCTION
www.tiem.utk.edu
Glycolysis, Citric Acid Cycle
www.thoughtco.com