What are some examples of liquids that evaporate?
Best Answers
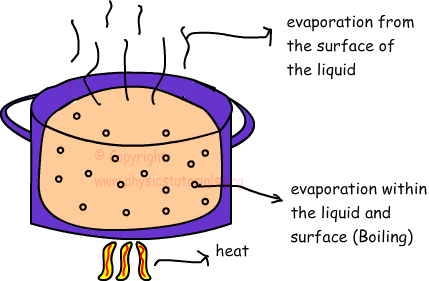
All liquids evaporate; meaning, at the surface of all liquids, there are some molecules that have a tendency to escape to become a gas. The question is, which liquids have surface molecules with the HIGHEST tendency to evaporate? read more
All liquids will evaporate. Some very quickly, some very slowly. Every liquid has an evaporation point. Higher temperature will generally speed up evaporation as well as lower pressure, See Boyle’s Law. read more
Liquids can boil, evaporate, freeze, condense and form solutions. The particles in water are free to flow, but viscosity may prevent some thick liquids from flowing. A high-viscosity liquid, such as honey, does not flow as quickly as a low-viscosity liquid, such as water. read more
Evaporation and condensation occur as matter changes physical states. Although they are explained as scientific processes, many examples of evaporation and condensation occur daily. Evaporation is the process of a substance in liquid form transitioning into the gas phase. read more
Encyclopedia Research
Related Questions
Related Facts
Related Types
Related Question Categories
Image Answers