What is the number of atoms in 0.004g of magnesium?
Best Answers
Mass of Mg=24g Number of moles in 0.004g=4/24000=1/6000 hence, number of atoms=(6.022*10^23)/6000 =6.022*10^20/6 3.011*10^20/3 10.026*10^19 atoms. read more
The number of atoms in 0.004g of magnesium is closed to (atomic mass of Mg =24 - 4179059 read more
Source: brainly.in
Encyclopedia Research
Wikipedia:
Related Questions
Related Facts
Related Types
Image Answers
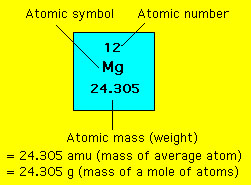
Source: learnnext.com
Further Research
How many atoms are present in 48.60 g of Mg?
socratic.org
How many atoms are there in 0.36g of magnesium?
socratic.org
The Number of atoms in 0 004g of Mg will be
www.meritnation.com