Why do atoms form bonds?
Best Answers
When this force of attraction brings atoms together to form substances containing two or more atoms, the bond is called a chemical bond. There are many types of chemical bonds, but the three major, or primary, types are ionic, covalent and metallic. read more
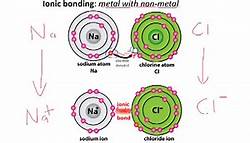
Atoms form chemical bonds in order to make their outer electron shells more stable. The type of chemical bond maximizes the stability of the atoms that form it. An ionic bond, where one atom essentially donates an electron to another, forms when one atom becomes stable by losing its outer electrons and the other atoms become stable (usually by filling its valence shell) by gaining the electrons. read more
Bonds are attractions between atoms. They usually share electrons, or gain them, or lose them to make sure their layers have the full electrons that they need. read more
Encyclopedia Research
Related Questions
Related Facts
Related Types
Image Answers