Why was Thomson's atomic model discarded?
Best Answers
Thomson based his theory on describing about the position of electron as only electrons were the fundamental particles discovered by his time. read more
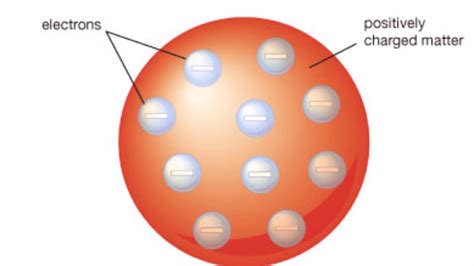
In 1888, Thomson suggested a model of atom. He proposed that the atom was spherical in shape and in that positive charges were uniformly distributed in this sphere of atomic dimension. Sufficient number of electrons were supposed to be embedded in it to balance the positive charge. read more
Thomson atomic model, earliest theoretical description of the inner structure of atoms, proposed about 1900 by William Thomson (Lord Kelvin) and strongly supported by Sir Joseph John Thomson, who had discovered (1897) the electron, a negatively charged part of every atom. read more
This model was likened to an Englishdessert called a plum pudding, analogizing electrons with theraisins randomly scattered through the interior of the pudding.This model of atomic structure was largely discarded in 1911, basedon discoveries made by Sir Ernest Rutherford. read more
Related Questions
Related Facts
Related Types
Related Question Categories
Image Answers